A new injectable sensing fibre was proposed and developed by a research team from Fudan University. The new sensing fiber performs well in electrochemical stability, mechanical properties, biocompatibility and biointegration, which makes long-term and real-time in vivo monitoring of the multiple chemicals possible.
The research, Functionalized helical fibre bundles of carbon nanotubes as electrochemical sensors for long-term in vivo monitoring of multiple disease biomarkers was published in Nature Biological Engineering on October 28. The research team was led by Peng Huisheng and Sun Xuemei from Department of Macromolecular Science, Yu Hongbo, School of Life Sciences, and Xu Fan, Department of Aeronautics and Astronautics.
With the development of medical technology and the increasing awareness of disease prevention, people have been paying more attention to the personalized medicine, like the real-time in vivo monitoring of multiple disease biomarkers. Electrochemical biosensor, a device used to transform chemical signals into electrical signals, can be used to detect specific chemicals and can be applied to wearable and implantable medical devices.
The existing in vitro electrochemical sensors work through sweat or urine. The above approaches, due to intermittent availability, simple types of components, lead to problems with continuity, accuracy and diversity of monitoring. In contrast, implantable sensors work through body fluids. They can realize accurate and real-time monitoring of chemicals in vivo and provide timely physiological information.
However, the majority of the existing implantable sensors have large moduli. With tissues’ movements, hard devices will damage soft tissues seriously and repeatedly. In addition, the existing implantable devices, designed based on a two-dimensional structure and implanted mainly by traditional surgeries, fail to realize mini-invasive implantation, thus resulting in the mechanical mismatches between implanted electronics and biological tissues as well as unsustainable and inaccurate monitoring. This affects signal acquisition and biosafety, which limits the sensors’ application.
To solve the problem, the research team of Fudan University designed an electrochemical sensor with helical fibre bundles that mimic the hierarchical structure of muscle. Firstly, the mechanics simulation and nano-indentation experiments show that carbon nanotube (CNT) fiber has lower bending stiffness than traditional implant materials such as gold wire or carbon fibre, and displays ultralow stress under compression. Secondly, drawing on the traditional intravenous injection method suitable for the one-dimensional structure of the fiber, the research team accurately injected the sensing fiber into the target area. The fiber after injection is similar to animal hair on the skin surface. Finally, the HE and immunofluorescence staining reveal the CNT fiber does not cause inflammation or scars in vivo after injection and is successfully integrated with the surrounding tissues. It proves that CNT fibre has excellent biocompatibility and biointegration.
Chemicals in vivo are manifold and unevenly distributed. In order to meet the practical needs of monitoring, the research team designed hierarchical helical sensing fibers with axial or radial structure. The multiply sensing fibers (MSF) can detect the distribution of the same substance at different sites, such as the distribution of H2O2 in tumors, or different chemicals at the same site, such as the calcium ions and glucose in the blood. Taking the latter as an example, the MSF can conduct real-time monitoring of the calcium ions and glucose in blood vessels, which matches well with ex situ measurements of blood samples by traditional commercial methods. With the help of the integrated circuit, Bluetooth and APP, the system can conduct remote and real-time collection of physiological data. Finally and the sensing fibers work stably in the blood vessels for four weeks. This research provides a new prospect for future bioelectronics.
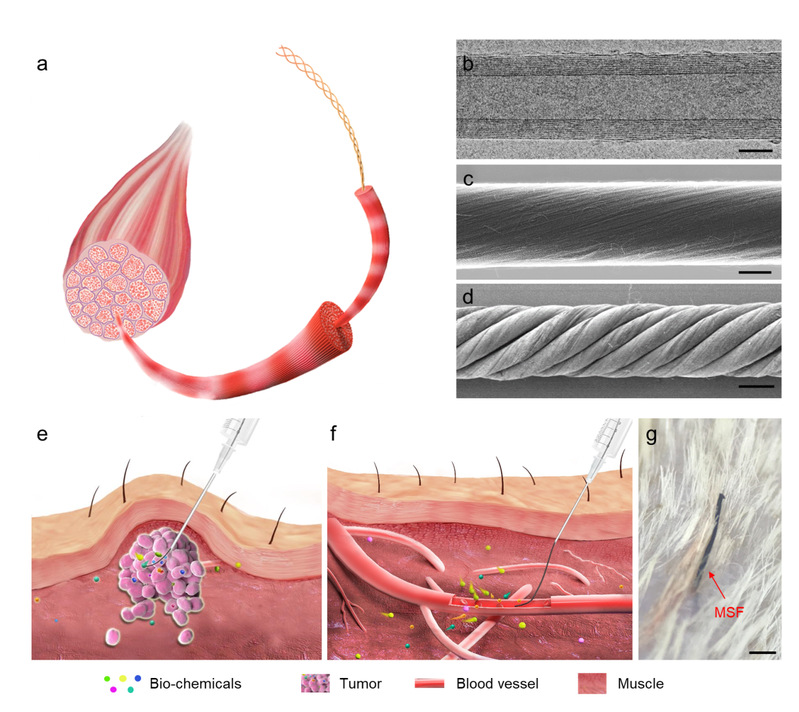
Fig. 1: Hierarchical helical structure and the implantation of CNT.
a.Schematic of the hierarchical structure of muscle.
b.Representative transmission electron microscope image of a multi-walled CNT.Scale: 3 nm
c&d. Representative SEM images of a primary CNT fibre (c) and a hierarchically helical CNT fibre assembled from primary CNT fibres (d). Scale bars, 6 μm (c) and 20 μm (d). e&f. Schematics showing the injection of the fibre into a target tumour (e) and blood vessel (f).
g. Photograph of the skin surface of a cat after injection with a MSF. Scale bar, 500 μm.e
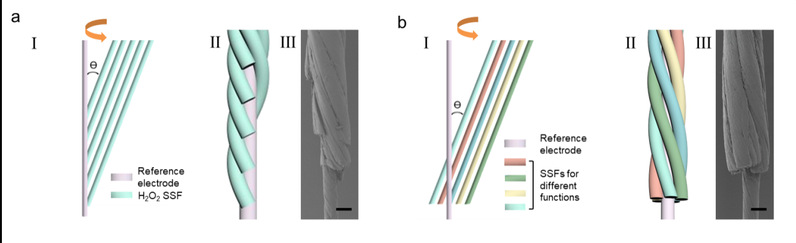
Fig. 2 a.: Schematic of the preparation process (i), schematic of the structure (ii) and a representative SEM image (iii) of MSFs. an MSF with axial terminals and five evenly spaced SSFs for spatial analysis.
b.an MSF with radial terminals for multiplex monitoring. Scale bar, 50 μm.





